Jul
24
The Miller-Urey Experiment Is Irrelevant
“We have reached a situation where a theory has been accepted as fact by some, and possible contrary evidence is shunted aside. [This is] mythology rather than science.” — Robert Shapiro, OOL researcher and gadfly to naturalist orthodoxy, Origins: A Skeptic’s Guide to the Creation of Life on Earth (1987)
You may have expected a post on current events this week, what with the RNC & DNC conventions, the latest shootings & bombings, controversial court rulings and legislation, etc. (Unfortunately, except for the conventions, the rest of it is all too common, these days.) But, I needed to focus on something else, so… let’s jump into some science, shall we?
 From what I’ve heard, it takes years for new scientific discoveries and theoretical breakthroughs to make it into the textbooks, particularly when they disrupt the preferred “scientific” narrative. After the initial papers are published in professional journals, the findings first show up in advanced, graduate-level books. Then, they appear in undergrad-level texts and, eventually, maybe, trickle down into High School and, finally, middle-school textbooks. Similarly, the popular-level science magazines and TV shows, while overhyping some discoveries, seem to forever repeat old theories, assumptions, and experiments, despite them having been disproven or, at least, cast into serious doubt by current research.
From what I’ve heard, it takes years for new scientific discoveries and theoretical breakthroughs to make it into the textbooks, particularly when they disrupt the preferred “scientific” narrative. After the initial papers are published in professional journals, the findings first show up in advanced, graduate-level books. Then, they appear in undergrad-level texts and, eventually, maybe, trickle down into High School and, finally, middle-school textbooks. Similarly, the popular-level science magazines and TV shows, while overhyping some discoveries, seem to forever repeat old theories, assumptions, and experiments, despite them having been disproven or, at least, cast into serious doubt by current research.
There doesn’t appear to be a good excuse for the delays, either. Seems to me, if the scientific establishment wanted to, they could pressure the writers, publishers, and producers to incorporate new discoveries and advances sooner. Heck, they might even sell more books that way. So, why not do this? Why do they drag their feet in informing students and the public? I’m not usually partial to conspiracy theories, and this may not even be a conscious effort on the part of many. But, it does occur to me that continuing to propagate old “knowledge” that supports metaphysical naturalism and/or evolution serves to keep the ignorant masses indoctrinated and intellectually invested in that idea, even once it has been discredited.
The iconic 1953 experiment by Miller and Urey is once such event that was invalidated decades ago, yet it is still taught as a significant discovery. Under the supervision of Nobel laureate Harold Urey, grad student Stanley Miller set out to test the Oparin-Haldane hypothesis. (Or, at least, part of it.) The idea was to reproduce the conditions of Earth’s primordial atmosphere and demonstrate how life might have arisen from nonliving chemicals. Prevailing theory at the time was that the Earth formed out of a condensing cloud of interstellar dust and gas (mostly hydrogen), so the primordial atmosphere would reflect that. Urey, Oparin, and Haldane all thought that the atmosphere would have been composed of hydrogen, methane, ammonia, and water vapor.
I should explain that this is an example of a “reducing” atmosphere, which is absent of free oxygen. A reducing atmosphere is required for the synthesis of organic molecules, as had been demonstrated in other experiments. On the other hand, an “oxidizing” atmosphere (i.e., one containing free oxygen) results in the process of respiration, in which energy is extracted from organic molecules by breaking them down. A reducing atmosphere was then assumed necessary for the appearance of first life and, thus, for Miller’s experiment.
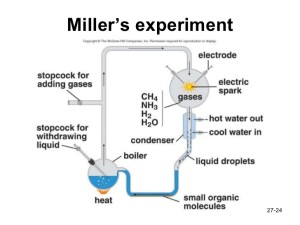 Miller built a closed gas apparatus (see pic) in Urey’s lab that would allow him to contain the above mixture (after pumping out the air), heat the water, circulate the gases past a sparking Tesla coil (i.e., to simulate lightning), and collect any resultant chemical residue. After a week, he sampled the now “deep red and turbid” water’s surface but found no amino acids. He rearranged his apparatus and tried again. This time, he discovered several organic compounds. Most of them were not the type found in living organisms. But, he did find the two simplest amino acids found in proteins — namely, glycine and alanine.
Miller built a closed gas apparatus (see pic) in Urey’s lab that would allow him to contain the above mixture (after pumping out the air), heat the water, circulate the gases past a sparking Tesla coil (i.e., to simulate lightning), and collect any resultant chemical residue. After a week, he sampled the now “deep red and turbid” water’s surface but found no amino acids. He rearranged his apparatus and tried again. This time, he discovered several organic compounds. Most of them were not the type found in living organisms. But, he did find the two simplest amino acids found in proteins — namely, glycine and alanine.
Afterward, Miller and others were able to reproduce the experiment. These efforts also yielded small amounts of “most [of the 20] biologically significant amino acids, as well as some additional organic compounds found in living cells.” (No one has ever been able to produce more glycine (2.1%) or alanine (1.7%) than Miller did in his second attempt.) The Miller-Urey experiment was deemed a successful confirmation of the Oparin-Haldane hypothesis and a breakthrough in itself. The results published in 1953 energized the scientific community, and a comprehensive theory of life’s origin was anticipated in the near future. Lay explanations in the popular press (e.g., New York Times, Life, Newsweek, Time) rocketed Miller to fame in the rest of the world, too. Problem was, there was already some doubt about the foundational assumptions of the experiment.
Even as early as 1952, geochemist Harrison Brown began questioning the origin and composition of Earth’s original atmosphere. In the 1960s, geochemist Heinrich Holland and geophysicist Philip Abelson echoed Brown’s conclusions. Abelson also pointed out that a) ammonia absorbs ultraviolet radiation from sunlight and would be quickly destroyed by it, and b) the lack of organic molecules in the earliest rocks indicates that methane was not present in abundance. Similarly, Sidney Fox and Klaus Dose later conceded that most of the hydrogen would have been lost to space, and the remaining methane and ammonia would likely be oxidized.
Abelson and Brown proposed instead that the early atmosphere was the result of volcanic outgassing, composed primarily of water vapor, carbon dioxide, nitrogen, and trace amounts of hydrogen. This is what is called a chemically “neutral” atmosphere. Biologists, geologists, and atmospheric scientists also chimed in over the next couple decades, with theoretical models implying some primitive oxygen (how much was uncertain) and biochemical evidence seeming to suggest significant levels of oxygen produced by the photo-dissociation of water vapor.
 In the late-70s to early-80s, a few origin-of-life (OOL) researchers began admitting that reluctance to give up the “reducing atmosphere” theory was because everyone well knew that the presence of even minute amounts of oxygen tends to shut down prebiotic chemical pathways. Geophysicists Harry Clemmey and Nick Badham declared it mere “dogma” to cling to the old theory in the face of actual, physical evidence to the contrary. (They were right.) This is particularly odd, though, since labs had been synthesizing organic molecules for many years, including in local environments with extremely low levels of oxygen. As Wells points out,
In the late-70s to early-80s, a few origin-of-life (OOL) researchers began admitting that reluctance to give up the “reducing atmosphere” theory was because everyone well knew that the presence of even minute amounts of oxygen tends to shut down prebiotic chemical pathways. Geophysicists Harry Clemmey and Nick Badham declared it mere “dogma” to cling to the old theory in the face of actual, physical evidence to the contrary. (They were right.) This is particularly odd, though, since labs had been synthesizing organic molecules for many years, including in local environments with extremely low levels of oxygen. As Wells points out,
“The success of the Miller-Urey experiment doesn’t prove that the entire primitive atmosphere lacked oxygen any more than the success of modern organic chemistry proves that the modern atmosphere lacks oxygen.”
I mentioned the problem of the lost hydrogen earlier. Hydrogen gas accumulates during Miller-Urey-type experiments, reaching up to 76% of the mixture, which does not mimic natural processes. Thus, Fox and Dose (1977) showed that, not only did Miller-Urey have the wrong “primordial atmosphere”,
“[it did] not satisfactorily represent early geological reality because no provisions [were] made to remove hydrogen from the system…. The inference that Miller’s synthesis does not have a geological relevance has become increasingly widespread.”
As more OOL researchers began to concede, more attention was paid to Abelson & Brown’s “neutral” theory. However, Fox and Dose reported that no amino acids were produced by performing a Miller-Urey-type experiment with water vapor, carbon dioxide, nitrogen, and a bit of hydrogen. Holland reported the same results. In a 1983 experiment with carbon dioxide, carbon monoxide, and a little free hydrogen, Miller and a colleague were only able to produce a small amount of glycine.
All the evidence indicates that prebiotic atmospheric conditions were hostile, not friendly, to the production of life’s building blocks. Exploring other OOL approaches, theories, and experiments is outside the scope of this article. The bottom-line, however, is that none have been able to compensate for the many crucial problems and develop a demonstrable scenario for purely naturalistic origins of life from non-life (aka abiogenesis). (See Origins of Life by Rana and Ross for a great discussion of the various bottlenecks and showstoppers for naturalistic scenarios.) It seems that life just can’t get started on its own, no matter how much time it has.
“[A]n accumulating body of geochemical evidence has shown that there likely never was a prebiotic soup. Two leading geochemists, James Brooks and Gordon Shaw, argued that if an ocean rich in amino and nucleic acids had existed, it would have left large deposits of nitrogen-rich minerals (nitrogenous cokes) in metamorphosed Precambrian sedimentary rocks. No evidence of such deposits exists. [And, as Brooks concluded,] ‘if such a soup ever existed it was only for a brief period of time.'” — Stephen C. Meyer, Signature in the Cell
 Ironically, as Rana and others have pointed out, Miller and those who followed him “demonstrated that this chemistry requires the input of an intelligent agent to successfully produce organic materials. Mechanistic studies highlight the need for careful adjustment of the gas composition and concentrations, selection of the right type of energy source, and implementation of special features to isolate and protect the prebiotic products from their energy source once they have formed. In other words, apart from the experimenters’ handiwork, gas phase reactions would never produce organic compounds.” Sounds like intelligent design rather than chemical evolution to me.
Ironically, as Rana and others have pointed out, Miller and those who followed him “demonstrated that this chemistry requires the input of an intelligent agent to successfully produce organic materials. Mechanistic studies highlight the need for careful adjustment of the gas composition and concentrations, selection of the right type of energy source, and implementation of special features to isolate and protect the prebiotic products from their energy source once they have formed. In other words, apart from the experimenters’ handiwork, gas phase reactions would never produce organic compounds.” Sounds like intelligent design rather than chemical evolution to me.
In Icons of Evolution (2000), Jonathan Wells noted several then-recent examples of publications that still promoted the idea that Miller-Urey successfully demonstrated the beginnings of organic life from inorganic compounds within a reducing atmosphere. These included the March 1998 issue of National Geographic, the 2000 edition of Ken Miller & Joe Levine’s Biology (a popular HS textbook), the 1998 Life: The Science of Biology (an undergrad textbook by Purves, Orians, Heller, & Sadava), the 1998 edition of Futuyma’s Evolutionary Biology (an advanced college textbook), the latest edition of Alberts et al.’s Molecular Biology of the Cell (a graduate-level textbook), and a 1999 booklet put out by the National Academy of Sciences (the U.S.’s premier science organization). Not much has changed since then.
Researchers and other scientists are justified in recognizing Miller-Urey for launching (sparking?) origin-of-life research as a formal scientific discipline. But, as should be clear, the assumptions and conclusions of the experiment itself have been proven to be grossly mistaken and effectively irrelevant. The question remains, however, “Why do the educators and popularizers of science continue to teach mythology?”
—
Sources:
Wells, Jonathan, Icons of Evolution (2000)
Rana, Fazale and Ross, Hugh, Origins of Life (2004)
Meyer, Stephen C., Signature in the Cell (2009)
Rana, Fazale, Creating Life in the Lab (2011)















